MiniPDX® in the Clinic: Guiding Treatment Decisions in 7 Days
Traditional PDX models can take 4–6 months, far too long to inform time-sensitive clinical decisions. MiniPDX® offers a clinically viable alternative, delivering drug-sensitivity data from patient-derived tumors in just 7 days, enabling oncologists to compare multiple regimens quickly and with high clinical correlation. This platform is especially impactful when standard therapies have failed or when treatment guidelines are unclear.
In China, MiniPDX® is a validated tool in the clinical setting, supported by multiple peer-reviewed publications. Its benefits to patients have led to a national Real-World Evidence (RWE) project approved by the Chinese Ministry of Industry and Information Technology. Through this initiative, a consortium of top hospitals and KOLs, including over 60 centers across China, will apply MiniPDX® to guide personalized treatment with a goal of reaching 10,000 cancer patients.
Below are examples of real patients who benefited from MiniPDX®-guided treatment
Unlocking New Treatment Options (Endometrial Stromal Sarcoma)
After 1st- and 2nd-line treatments failed, a patient with metastatic endometrial stromal sarcoma received MiniPDX® testing. Surprisingly, the best responder was apatinib, which is conventionally used as 3rd line treatment for gastric cancer. Based on the MiniPDX® results, the patient began apatinib therapy. Followup CT scans show near complete resolution of lung metastases.
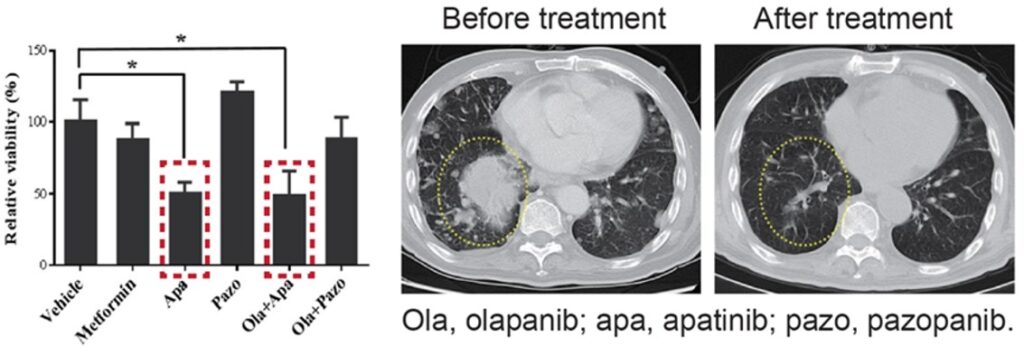
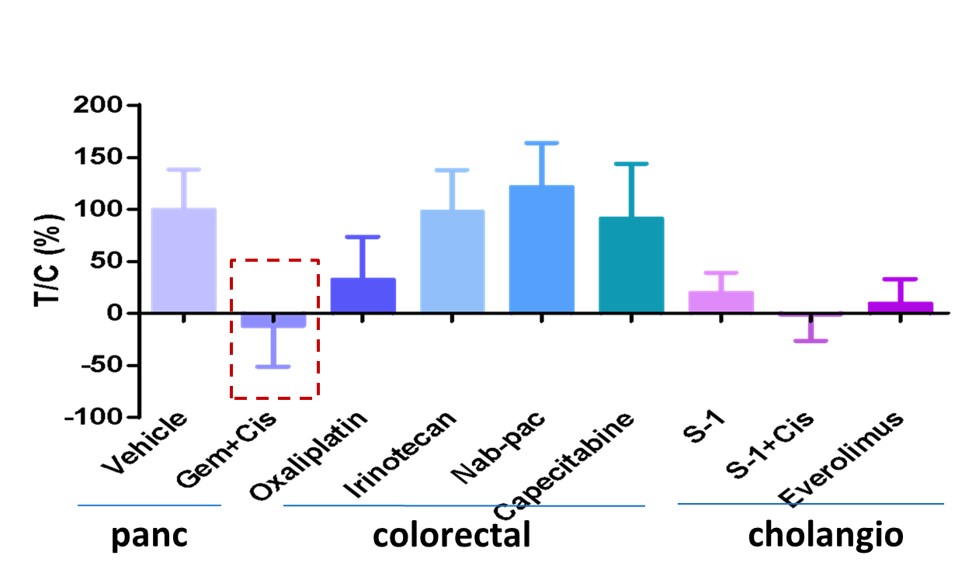
Tailoring Therapy in Rare Cancers (Periampullary Tumor)
A patient with a duodenal periampullary tumor-an ultra-rare cancer with no standard regimen-underwent MiniPDX® testing. There is a wide selection of conventional treatment options, guided by related cancers pancreatic, colorectal or cholangio. Traditional treatment pathways would start with colorectal cancer therapies but MiniPDX® results guided gemcitabine treatment for this patient, a pancreatic cancer agent. The patient responded very well, underscoring MiniPDX®’s value in rare and ambiguous cases.
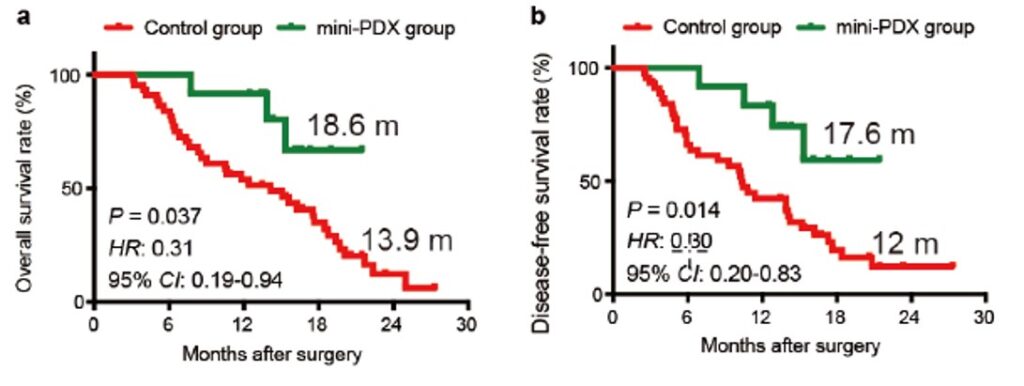
Extending Survival (Gallbladder & Gastric Cancer)
A landmark study by Shanghai Ruijin Hospital compared MiniPDX®-guided therapy to standard-of-care in gallbladder carcinoma patients. Those guided by MiniPDX® experienced an average 5-month improvement in both overall survival (OS) and disease-free survival (DFS). Similar results were reported by Nanjing First Hospital in gastric cancer (GCLM) patients.