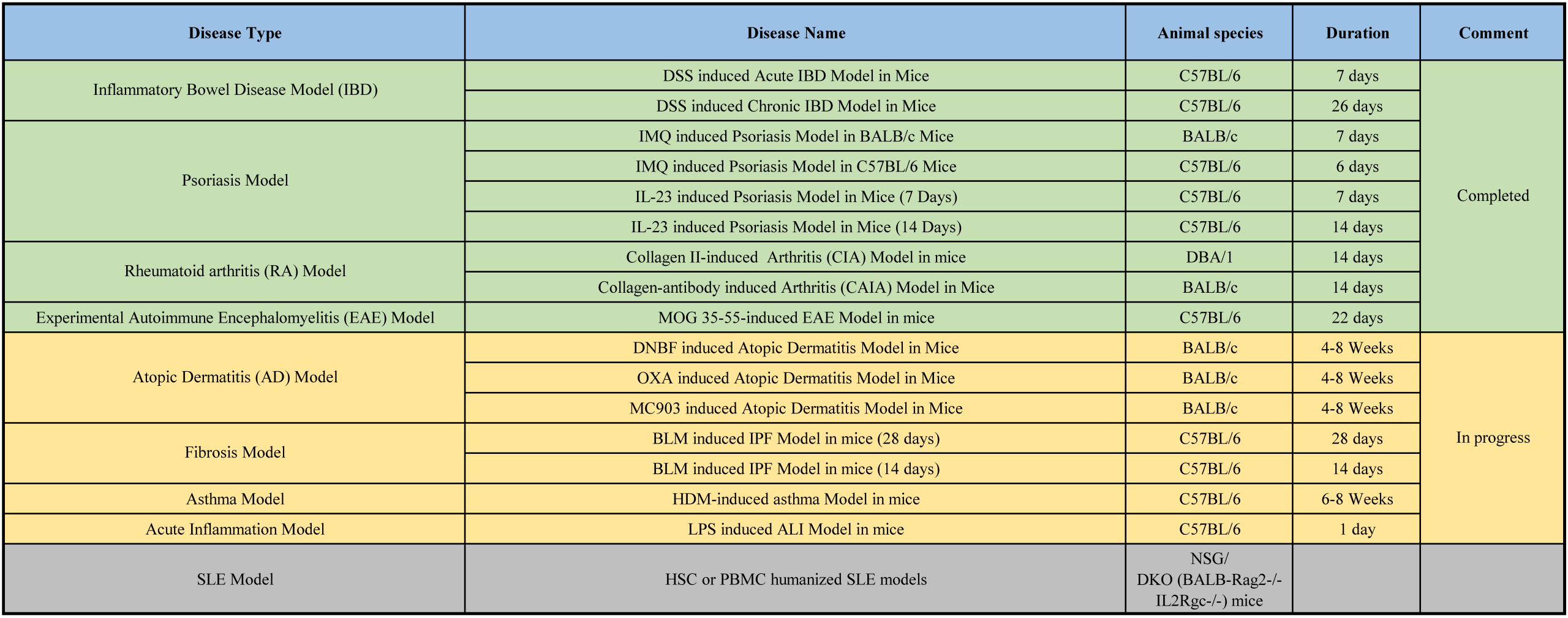
Autoimmune Disease Models
LIDE provides a comprehensive platform of validated autoimmune disease models designed to accelerate your drug discovery and development programs. Our platform features highly reproducible models for Inflammatory Bowel Disease (IBD), Psoriasis, and Rheumatoid Arthritis (RA), offering both acute and chronic options to match your specific therapeutic mechanism.
Our Core Autoimmune Disease Platforms
- Inflammatory Bowel Disease (IBD) Models
We utilize Dextran Sulfate Sodium (DSS) to induce colitis, mimicking the clinical and histopathological features of human ulcerative colitis.- DSS-Induced Acute Colitis: A 7-day model focused on innate immunity dominance (macrophages and dendritic cells).
- DSS-Induced Chronic Colitis: A 26-day recurrent model that involves adaptive immunity (Th1/Th17) and allows for the study of fibrosis.
- Key Readouts: Disease Activity Index (DAI) scoring (weight loss, stool consistency, blood), colon length measurement, and histopathological analysis of mucosal damage and infiltration.
- Psoriasis Models
- Our psoriasis platform offers multiple induction methods to evaluate therapies targeting the IL-23/IL-17 axis
- IMQ-Induced Models (BALB/c or C57BL/6): Utilizing topical Imiquimod application to induce severe (BALB/c) or (C57BL/6) lesions within 6-7 days.
- IL-23 Induced Models: Direct intradermal injection of recombinant IL-23 into the ear, available in 7-day and 14-day protocols.
- Key Readouts: PASI clinical scoring (erythema, scaling, thickness), ear thickness measurement, spleen index, histopathological analysis and serum cytokine analysis.
- Rheumatoid Arthritis (RA) Model
- We offer the Collagen-Antibody Induced Arthritis (CAIA) model for rapid evaluation of anti-inflammatory candidates.
- CAIA Model: A 14-day passive immunity model induced by an antibody cocktail and LPS enhancement
- Key Readouts: Clinical arthritis scoring, paw thickness measurements, and histopathology.
Comprehensive Study Capabilities
Every study at LIDE is supported by a standardized, high-quality workflow to ensure data integrity and clinical relevance.
| Capability | Details |
| Animal Strains | Validated in C57BL/6J and BALB/c mice. |
| Dosing Routes | P.O., I.V., I.P., S.C., and Topical applications. |
| Clinical Monitoring | Daily body weight, specialized scoring (DAI, PASI), and physical measurements. |
| Endpoints | Histopathology (H&E), serum cytokine analysis (ELISA/Multiplex), and tissue collection. |
| Standard of Care | Benchmarked against positive controls like Cyclosporine A, Deucravacitinib (BMS-986165), and Dexamethasone. |
Why Partner with LIDE?
- We help you “pick the model” based on your drug’s specific mechanism—whether it targets innate vs. adaptive immunity or acute vs. chronic inflammation.
- Data-Driven Precision: Our reports include robust statistical analyses (ANOVA, t-tests) and high-resolution histopathology to provide a clear picture of therapeutic efficacy.