MiniPDX® – the 7 Day In Vivo 3D Organoid
LIDE’s MiniPDX® combines the predictive power of traditional PDX with a rapid 7‑day turnaround, offering a scalable in-vivo assay that preserves clinical relevance.
It is well known that patient-derived xenografts (PDXs) represent the gold standard in preclinical drug efficacy validation. LIDE has one of the largest PDX model libraries in the world. However, long lead times to expand PDX models and tight budgets have limited PDX assays to only the most promising targets.
MiniPDX® is a faster, cheaper alternative to PDX studies, without a loss in predictive power for drug response.
Comparison of PDX, Organoids and MiniPDX
| PDX | MiniPDX | 3D Spheroid / Organoids | |
|---|---|---|---|
| Study Type | In-vivo | In-vivo | In-vitro |
| Study Length | 4-6 months | 7-10 days | 2-8 weeks |
| Cost | High | Med | Low |
| Predictive Power | 89% | 82%* | TBD |
| Primary Benefit | Gold standard validation for IND application – well characterized | Predictive power of PDX but shorter turnaround time. Better representation of natural tumor environment than 3D or 2D cell cultures | Better representation of natural tumor environments than 2D cell cultures |
*by calculation here
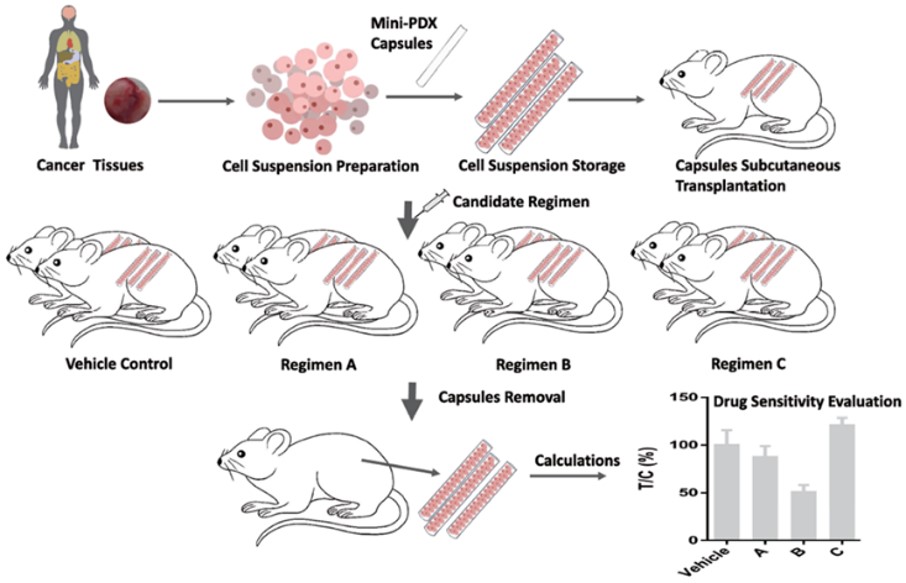
- Sample Preparation: Use fresh patient tumors or existing PDX tissue. Dissociate into cell suspensions.
- Encapsulation: Load cells into MiniPDX capsules with 500 kDa permability, allowing free flow of most drugs while maintaining cancer cells within the tube membrane.
- Implantation and Dosing: The capsules are implanted subcutaneously, 2 capsules per mouse, one in each flank, followed by systemic drug administration over 7 days. Specific regimens can be client-modified and can be delivered orally or via injection.
- Rapid Analysis: Retrieve capsules and assess cell viability.
- Clinical Correlation: 92% positive predictive value compared to standard PDX models.
Read the paper › - Mice Reduction: Each study requires 70% less mice than an equivalent PDX study.
See our 2021 3Rs award › - Less Compounds: A shorter dosing window means more precious compounds for other studies.
- In Vivo Applicability: Captures real pharmacokinetics and metabolism of the drug in a whole animal
- Timeline Savings: Results in as little as 10 days instead of the 4-6 months needed for a PDX study.
- Budget Efficiency: 10-25% the cost of a comparable PDX study. Request a quote ›
Several papers have been published supporting MiniPDX®