MiniPDX® for Immuno Oncology: IO-FIVE®
IO-FIVE™ (Immuno-Oncology Fast In Vivo Efficacy) is LIDE’s advanced adaptation of our proven MiniPDX® platform, purpose-built for immuno-oncology drug candidates. Using fresh patient tumor samples and humanized immune-reconstituted mice, IO-FIVE™ delivers actionable in vivo efficacy data for IO agents in as little as 14 days.
How IO-FIVE® Works:
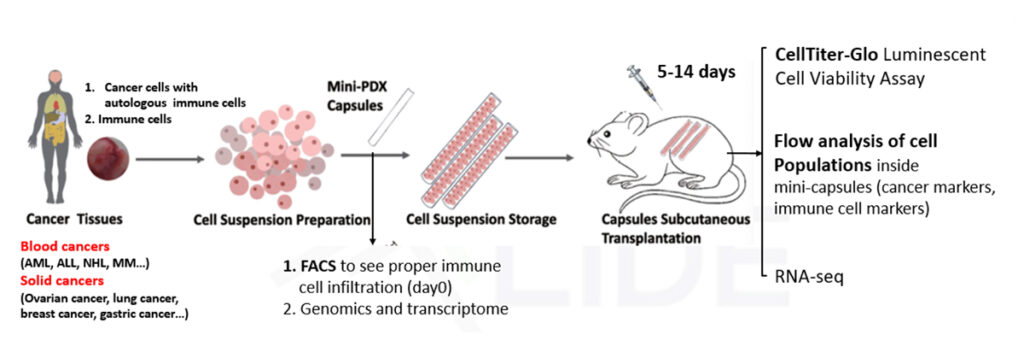
- Fresh Clinical Sample Acquisition: Tumor biopsies or malignant effusions from cancer patients are collected and processed immediately.
- Cell Dissociation: Patient tumors are digested and dissociated into cell suspensions.
- MiniPDX®-based Implantation: Mini-capsules containing a mixture of autologous immune cells and tumor cells are implanted into mice. 2 capsules per mouse.
- Therapeutic Treatment: Systemic dosing of implanted mice
- Endpoint Analysis – within 14 days, IO-FIVE™ provides:
- Tumor cell viability reduction
- Immune cell activation and infiltration
- Multiparameter FACS immune phenotyping
- Cytokine release profiling
- Speed: Results in 14 days, enabling faster go/no-go decisions in IO programs.
- Clinical Relevance: Uses fresh patient tumors and matched immune components for high translational value.
- Immune Profiling Depth: Comprehensive immune phenotyping (CD45, CD3, CD4/CD8, activation/exhaustion markers) and cytokine analysis.
- Versatility: Compatible with solid tumors, hematologic malignancies, and difficult-to-model rare cancers.
- Integration: Seamlessly connects with LIDE’s humanized PDX, co-culture assays, and omics platforms for end-to-end IO development.