Syngeneic Mouse Models – Fast & Immunocompetent
Syngeneic models remain a cornerstone for immuno-oncology (IO) drug development, offering the speed, cost-efficiency, and immunological integrity needed for early-stage evaluation. LIDE’s syngeneic and humanized syngeneic mouse platforms are designed to evaluate immune checkpoint inhibitors, cytokine modulators, bispecific antibodies, cell therapies, and combination regimens in fully immunocompetent hosts.
| Advantage | Impact on R&D |
|---|---|
| Fully functional immune system | Enables evaluation of immune-mediated antitumor effects |
| Rapid tumor growth | Short study timelines accelerate go/no-go decisions |
| Reproducible and scalable | Consistent tumor take rates for high-throughput IO screening |
| Broad tumor repertoire | Solid and hematologic malignancies represented |
| Compatible with genetic engineering | Allows knock-in/knock-out models for human target evaluation |
LIDE’s Syngeneic Model Portfolio
We maintain a diverse collection of murine tumor cell lines covering solid, blood, and orthotopic models. All models are available with or without bioluminescent variants for real-time, non-invasive tumor monitoring.
- Brain: GL261
- Breast: 4T1 (subcutaneous & mammary fat pad), EMT-6
- Colorectal: CT26.WT, MC38
- Gastric: MFC
- Kidney: RAG, Renca
- Liver: H22, Hepa1-6
- Lung: LLC1 (LL/2)
- Pancreas: PAN02
- Prostate: RM-1
- B-cell Lymphoma: A20
- T-cell Lymphoma: E.G7-OVA
- Melanoma: B16-F10
- MBT2
Orthotopic Syngeneic Models:
LIDE also offers orthotopic syngeneic models for greater clinical relevance by implanting tumors into their organ of origin. Available models include:
- Liver: Hepa1-6-luc, H22-luc
- Breast: 4T1-luc (triple-negative)
- Pancreas: Panc02-luc
- Bladder: MB49-luc
Humanized Syngeneic Models
For evaluating human-specific targets in an immunocompetent setting, we offer huPD-1 or huPD-L1 knock-in syngeneic models. This is perfect for testing antibodies like pembrolizumab or nivolumab, which do not cross-react with murine PD-1.
Model Customization Available
If your study requires a specific indication, strain, or delivery route, LIDE’s scientific team can support custom syngeneic model development on request.
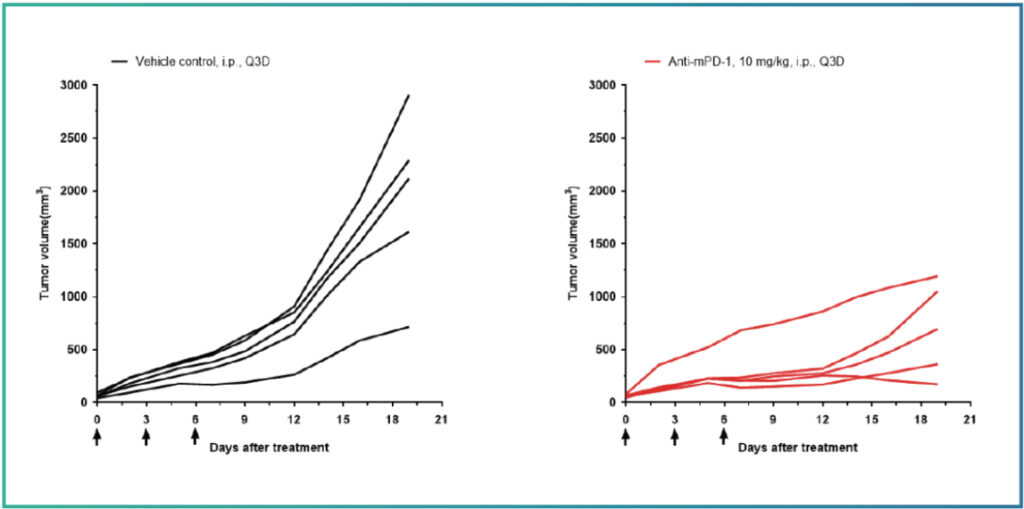
Example Study:
Drug efficacy analysis in EMT-6 subcutaneous transplantation model after administration of anti-mPD-1 therapy. All 5 mice exhibited partial response. TGI (tumor growth inhibition) rate was 66.16%.