Accelerating ADC Development
Antibody-drug conjugates (ADCs) represent one of the fastest-growing therapeutic classes in oncology. Their complexity-antibody specificity, linker stability, payload potency, and bystander effects-requires preclinical platforms that reflect both efficacy and safety in clinically relevant systems.
LIDE offers a unique combination of patient-derived models, rapid MiniPDX® assays, and large-scale mouse clinical trials to help ADC programs move efficiently from discovery to the clinic.
Why ADC Development Needs More Than Traditional Models
Conventional CDX or 2D in vitro systems fail to capture the tumor heterogeneity, antigen expression variability, and microenvironment interactions critical for ADC success. PDX and MiniPDX® models provide:
- Patient-matched antigen expression profiles
- Heterogeneous tumor environments that reveal off-target and bystander effects
- Biomarker discovery potential for patient stratification in trials
MiniPDX® – A Rapid ADC Evaluation Platform
Unlike small molecules, ADCs often show slower pharmacokinetics and distinct mechanisms of action. Traditional PDX studies take 4–6 months, delaying insights.
MiniPDX® solves this challenge:
- Uses PDX tissue or fresh patient tumors implanted into capsules and engrafted in mice
- Produces drug sensitivity data in ~7–10 days
- Well correlated with PDX and clinical outcomes (82% correlation to patient response)
- Ideal for screening payloads, linkers, and ADC formats quickly and cost-effectively
PDX Mouse Clinical Trials for ADCs
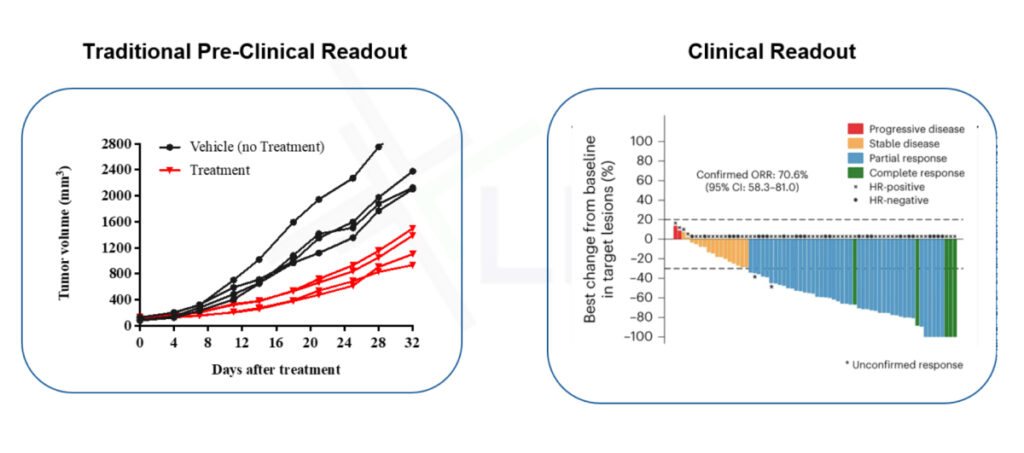
Traditional preclinical studies often rely on a handful of xenograft models, which may show strong activity in early tests but fail to reflect the heterogeneity of real patient populations. This gap is especially risky for ADCs, where antigen expression, tumor microenvironment, and resistance mechanisms vary widely.
Mouse Clinical Trials (MCTs) solve this problem by simulating the design and diversity of a human clinical trial-using dozens to hundreds of clinically annotated PDX models to generate robust, translational datasets.
Advantages of Mouse Clinical Trials
Clinical Relevance
- Large PDX cohorts capture the heterogeneity of patient populations, including rare genotypes and resistant tumors.
- Allows evaluation of ADC efficacy across variable antigen expression levels and microenvironments.
- MCT data are increasingly cited in IND packages as supportive evidence of efficacy and biomarker rationale.
Trial-Like Design
- MCTs mimic actual clinical trial structures (e.g., basket trials, umbrella trials, combination regimens).
- Data are reported as response rates, survival curves, and biomarker-linked efficacy metrics, directly translatable to clinical endpoints.
Biomarker Discovery
- By pairing PDX results with genomics and proteomics, MCTs help identify predictive biomarkers to guide patient selection.
- Supports regulatory submissions by showing evidence of stratification strategies before human trials.
Resistance & Combination Strategies
- PDX models that are naturally resistant to chemotherapy, targeted therapies, or immunotherapies provide insight into how ADCs perform in challenging real-world scenarios.
- Enables rational design of combination regimens to overcome resistance.
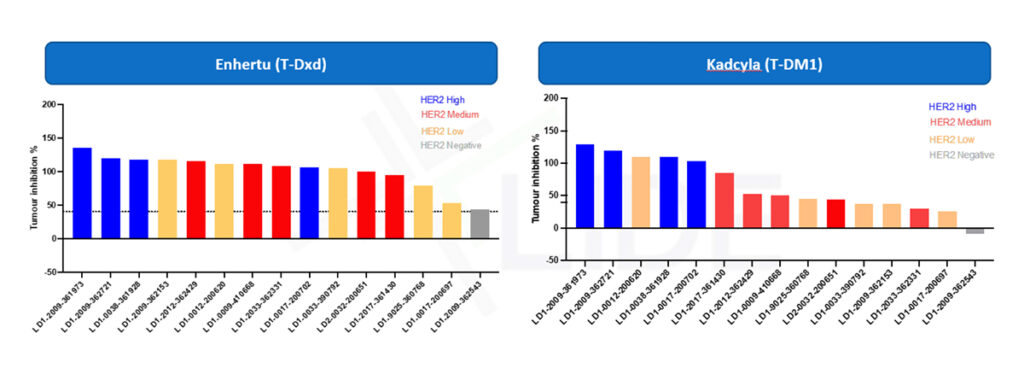
Example Study: Enhertu vs Kadcyla tumor inhibition across different HER2 expression levels